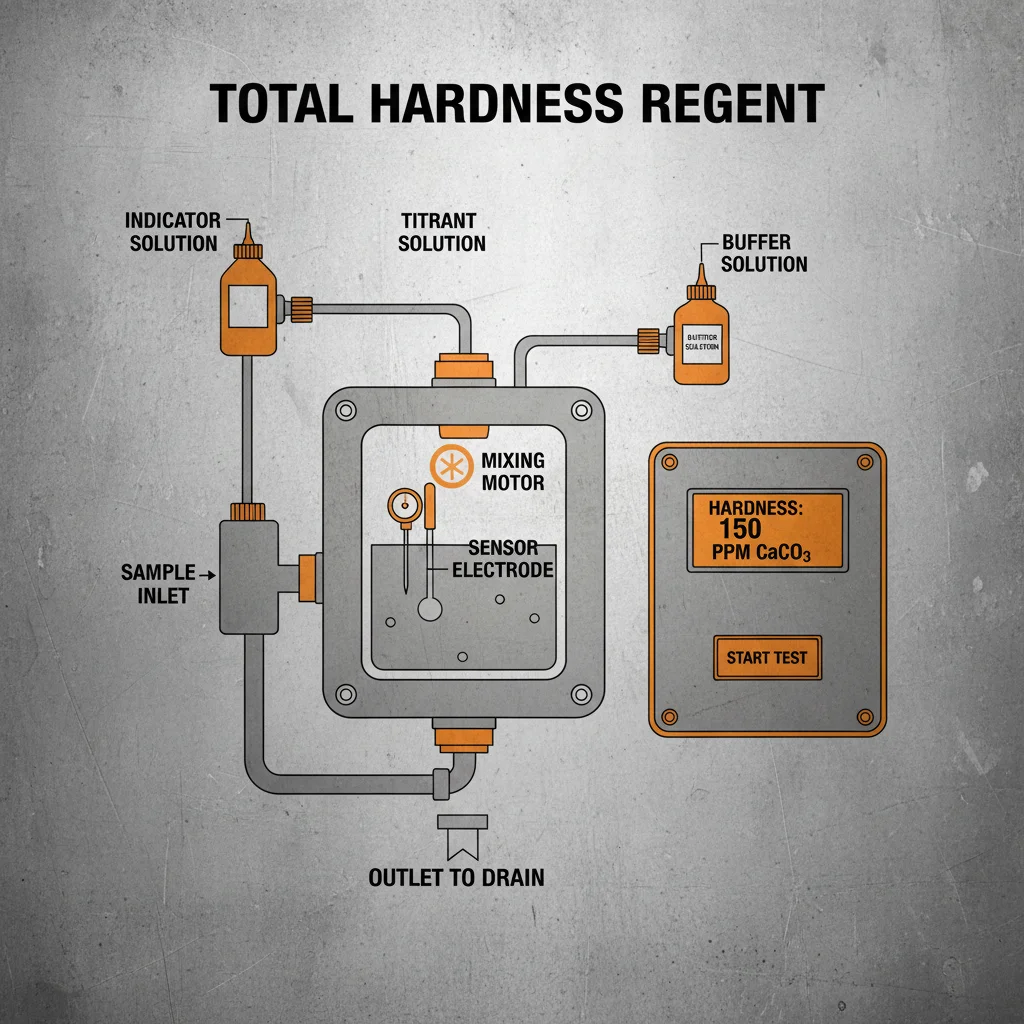
Understanding Alkalinity Analyzers: A Comprehensive Guide
Maintaining optimal water chemistry is crucial across various industries, from wastewater treatment to aquaculture and even home brewing. A key parameter in this process is alkalinity – the water’s capacity to neutralize acids. An alkalinity analyzer provides accurate and reliable measurements of this vital characteristic. This article explores the importance of alkalinity testing, the different types of analyzers available, their applications, and factors to consider when selecting the right instrument for your needs. Accurate alkalinity measurement ensures efficient process control and compliance with regulatory standards.
Why is Alkalinity Measurement Important?
Alkalinity acts as a buffer, resisting changes in pH. Consistent pH levels are critical for biological processes, chemical reactions, and overall system stability. In wastewater treatment, proper alkalinity control optimizes coagulation and disinfection. In aquaculture, it ensures a healthy environment for aquatic life. Monitoring alkalinity also helps prevent corrosion in pipelines and industrial equipment. Without accurate alkalinity measurements, processes can become unstable, leading to inefficiencies, environmental issues, and increased operating costs. Therefore, investing in a reliable alkalinity analyzer is a proactive step towards ensuring operational efficiency and environmental responsibility.
Key Benefits of Alkalinity Monitoring: Optimize process control, prevent corrosion, maintain stable pH, ensure regulatory compliance, and improve water quality.
Types of Alkalinity Analyzers
Several types of alkalinity analyzers are available, each with its own advantages and disadvantages. Titration-based analyzers are a traditional method, involving the controlled addition of acid to a water sample until a specific endpoint is reached. These are often manual, requiring skilled operators, but are relatively inexpensive. Electrometric analyzers utilize specialized electrodes to directly measure alkalinity, providing real-time, continuous monitoring. These are more expensive but offer greater automation and precision. Colorimetric analyzers employ chemical reactions that produce a color change proportional to the alkalinity concentration. These are simpler to operate than electrometric analyzers but may be less accurate. Choosing the right type depends on your budget, required accuracy, and level of automation.
Analyzer Type Comparison:
• Titration: Low cost, manual operation, requires skilled technician.
• Electrometric: Real-time monitoring, high accuracy, automated.
• Colorimetric: Simple operation, moderate accuracy, moderate cost.
Applications of Alkalinity Analyzers
The applications of alkalinity analyzers are widespread. In wastewater treatment plants, they ensure efficient neutralization of acidic or alkaline effluent before discharge. Power plants utilize them to monitor boiler water chemistry, preventing scale formation and corrosion. Aquaculture farms rely on accurate alkalinity measurements to maintain optimal conditions for fish and shellfish growth. Industrial processes involving pH-sensitive reactions benefit from continuous alkalinity monitoring. Furthermore, Jensprima’s alkalinity analyzers find applications in research laboratories, environmental monitoring, and drinking water treatment facilities.
| Industry |
Application |
| Wastewater Treatment |
Effluent Neutralization, Process Control |
| Power Generation |
Boiler Water Chemistry Monitoring |
| Aquaculture |
Water Quality Management |
| Industrial Processing |
pH Control, Reaction Optimization |
Choosing the Right Alkalinity Analyzer
Selecting the appropriate alkalinity analyzer requires careful consideration of several factors. Accuracy and precision are paramount, especially in critical applications. Consider the required measurement range and the type of sample you'll be analyzing. Ease of use and maintenance are also important, particularly for operators with limited training. Look for features like automatic calibration, data logging, and remote monitoring capabilities. Finally, consider the total cost of ownership, including initial purchase price, consumables, and maintenance expenses. Jensprima offers a range of analyzers designed to meet diverse needs and budgets.
Jensprima Alkalinity Analyzer Specifications
The Jensprima alkalinity analyzer offers a reliable and accurate solution for a wide range of applications. Here's a detailed overview of its key specifications:
| Specification |
Detail |
| Measurement Range |
0 – 2000 mg/L as CaCO3 |
| Accuracy |
±1% Full Scale |
| Resolution |
0.1 mg/L |
| Operating Temperature |
5 – 40°C |
| Power Supply |
100-240V AC, 50/60Hz |
Conclusion: Invest in Precise Alkalinity Measurement with Jensprima
Investing in a high-quality alkalinity analyzer from Jensprima is an investment in process efficiency, environmental compliance, and long-term cost savings. Our analyzers are designed for accuracy, reliability, and ease of use, ensuring you have the data you need to make informed decisions. Contact us today to learn more about our product offerings and find the perfect solution for your specific application.
Frequently Asked Questions (FAQs)
What factors can affect alkalinity measurements?
Several factors can influence alkalinity measurements. Temperature can affect the solubility of carbon dioxide, which impacts alkalinity. Sample contamination from acids or bases can also lead to inaccurate readings. The presence of certain ions, such as sulfates and chlorides, can interfere with some measurement methods. Proper sample collection and storage are crucial, as prolonged exposure to air can alter alkalinity levels. It’s also essential to calibrate the alkalinity analyzer regularly using certified standards to ensure accurate results.
How often should I calibrate my alkalinity analyzer?
The frequency of calibration depends on the analyzer type, usage intensity, and required accuracy. As a general guideline, calibrate your analyzer at least once a week, or more frequently if you are analyzing samples with varying alkalinity levels. Always calibrate after replacing any parts that affect the measurement process. Refer to the manufacturer's instructions for specific calibration recommendations. Jensprima analyzers are designed for ease of calibration, ensuring consistent and reliable results.
What is the difference between total alkalinity and individual alkalinity components?
Total alkalinity represents the overall capacity of water to neutralize acids. However, alkalinity is contributed by various components, primarily bicarbonate (HCO3-), carbonate (CO32-), and hydroxide (OH-). Individual alkalinity components provide a more detailed understanding of the water chemistry. Measuring these components can help identify the source of alkalinity and optimize treatment processes. A comprehensive alkalinity analyzer can often measure these components separately.
What are the best practices for sample collection for alkalinity testing?
To ensure accurate alkalinity measurements, follow these best practices: Collect samples in clean, inert containers (plastic or glass). Avoid exposing the sample to air for prolonged periods. Analyze the sample as soon as possible after collection. If immediate analysis is not possible, store the sample at 4°C to minimize changes in alkalinity. Filter the sample to remove any particulate matter that could interfere with the measurement. Always record the sample date, time, and location.




















































































 Download
Download Video Center
Video Center Technical Services
Technical Services FAQS
FAQS Service Network
Service Network Privacy Policy
Privacy Policy Contact Us
Contact Us






 LEARN MORE
LEARN MORE